Definition
adjective
(1) (used of solutions) Of or having the same or equal osmotic pressure.
(2) A condition in which the total number of solutes (i.e. permeable and impermeable) in a solution is the same or equal to the total solutes in another solution.
Supplement
Word origin: G. isos, equal + osmotic: relating to osmosis.
Related forms: isosmotically (adverb).
Related phrases: isosmotic solution.
Alternative forms: isoosmotic, iso-osmotic.
Compare: hyposmotic, hyperosmotic.
See also: osmosis, osmotic pressure.
Dictionary > Isosmotic
You will also like...
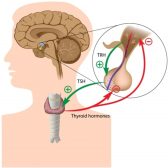
Hormone Production
Hormones are chemical messengers produced by specialized glands and they were produced by switching on the genes designe..

Ecology & Biodiversity: New Zealand Flora & Fauna
New Zealand is known for its unique biodiversity, caused by its remarkable geography and geologic history. Breaking away..

The Gene Pool and Population Genetics
According to Charles Darwin's theory of natural selection, preferable genes are favored by nature in the gene pool, and ..

Evolution of Life – Ancient Earth
Autotrophs flourished, absorbing carbon and light. Soon after, primitive life forms that could assimilate oxygen thrived..

Lotic Communities & Algae
Lotic communities have conditions that are rather harsh for typical plants. Thus, the diversity of plant species in loti..
Digestion and Absorption of Food
The gastrointestinal system breaks down particles of ingested food into molecular forms by enzymes through digestion and..

